Detection Methods for Lithium-ion Batteries Containing Ignition Causes
This page describes the conditions that might cause lithium-ion batteries to ignite, as well as the typical inspection methods used throughout the production process to discover cells with ignition factors.
Factors that can cause lithium-ion batteries to ignite
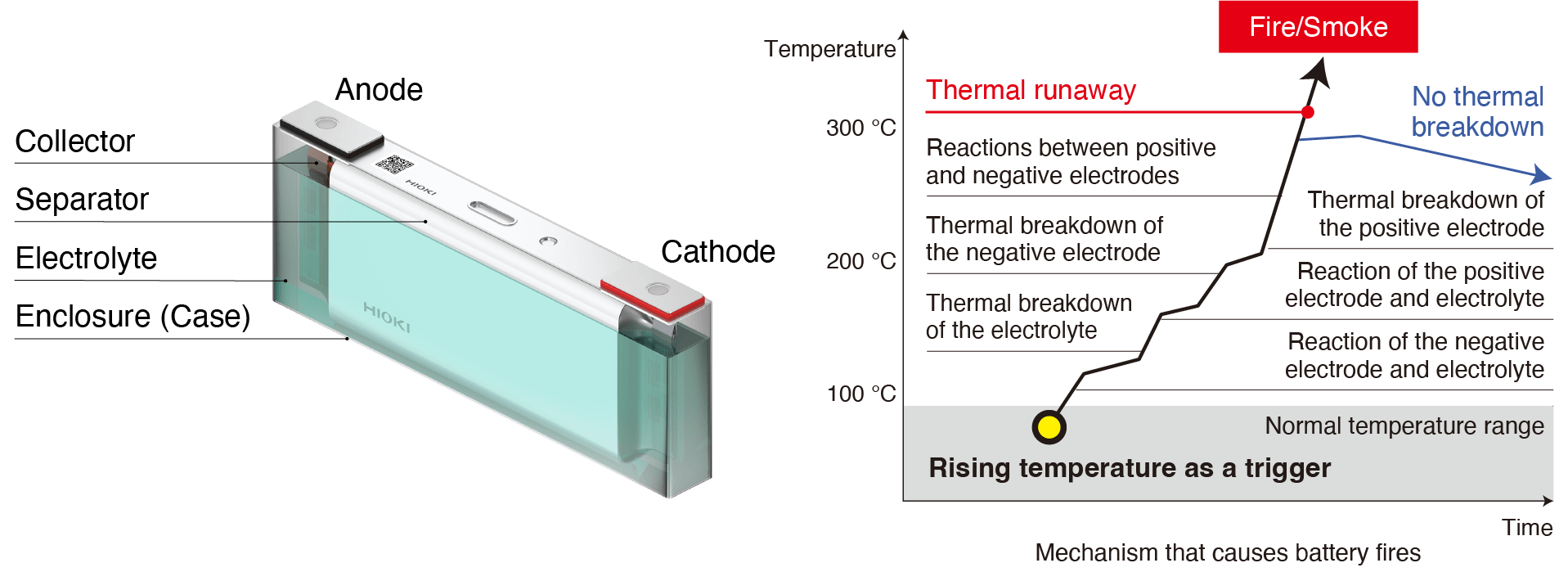
Broadly speaking, lithium-ion batteries consist of a cathode, anode, separator, electrolyte, collector, and enclosure. Structurally, their cathode and anode are insulated from each other by the separator. If this insulation is compromised - in other words, if a short-circuit occurs - the battery’s temperature will rise. Such a condition, known as a thermal runaway, can cause the battery to catch fire. Similarly, if the welds between materials are defective, repeated charging and discharging can cause the welds to heat up and ignite.
Factors that cause short circuits
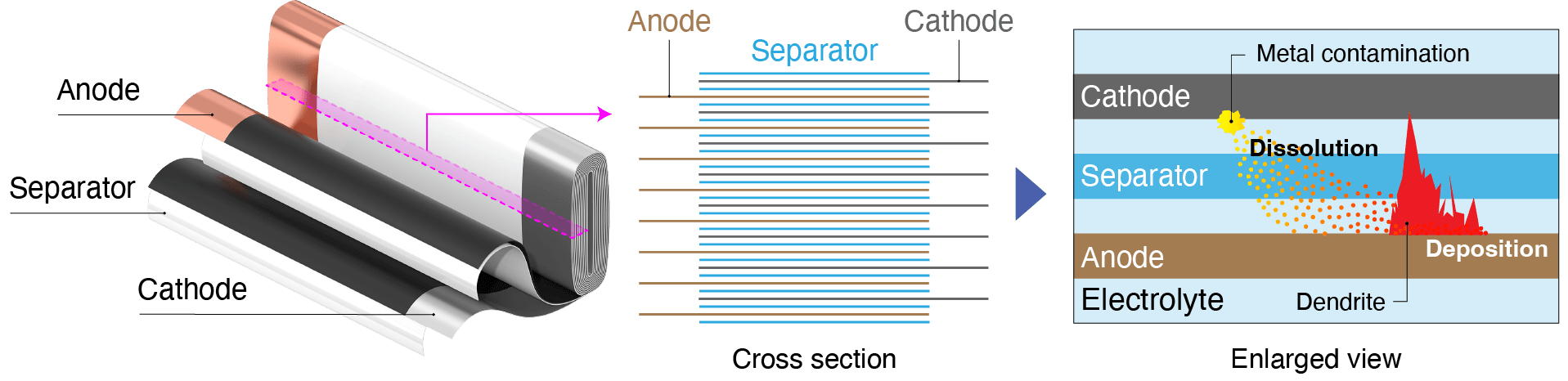
Short-circuits between the cathode and anode can be caused by factors including external impact, overcharging, material degradation, and contamination with minuscule quantities of metal. If present in a battery, such contaminants can gradually dissolve into the electrolyte and then precipitate to form tree-like deposits. Metal that has undergone this process is known as dendrite. Growth of dendrite over time can cause the separator to rupture, resulting in a short-circuit.
Detect batteries containing ignition factors
A variety of tests are carried out during battery manufacturing processes to detect batteries containing ignition factors. Most battery manufacturers use insulation resistance testing and open-circuit voltage testing for this purpose. These two types of testing play an essential role in the manufacture of safe batteries.
Insulation Resistance Testing
The cathode and anode in a lithium-ion battery are insulated from one another by the separator. This insulated state is generally verified by making judgments based on the resistance value between electrodes. Manufacturers verify that the insulation is good, which is to say, that the resistance value is high, and that current does not flow. It’s also necessary to keep each electrode insulated from the battery’s enclosure. As a result, insulation resistance testing is also performed between each electrode and the enclosure.
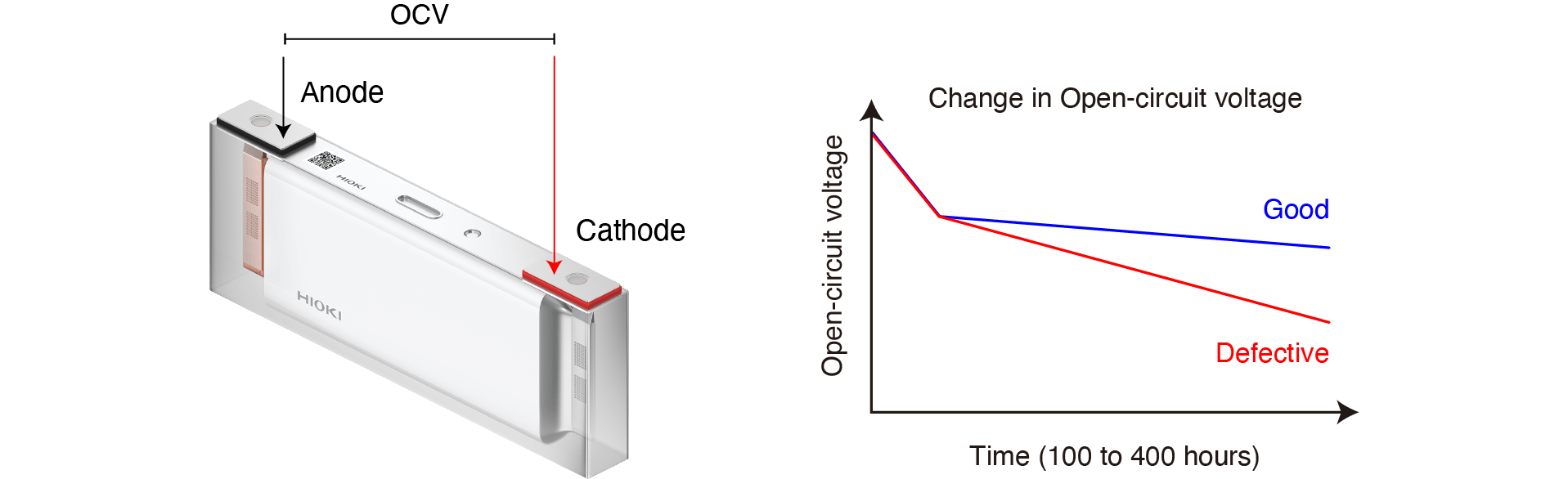
Open-circuit Voltage (OCV) Testing
The battery’s voltage when not connected to a load is known as the open-circuit voltage. Batteries exhibit self-discharge characteristics, which cause the open-circuit voltage value to gradually decline. When a short-circuit occurs, the self-discharge increases. Batteries whose open-circuit voltage exceeds a certain control value are classified as defective.